What is absolute entropy4/18/2024 
Standard absolute entropy is defined as the absolute entropy of a substance in its standard state that is, at atmospheric pressure 1 atm and temperature being 298.15 K or $$ because in solids, intermolecular forces are large and hence, atoms are less randomly distributed or possess negligible randomness. 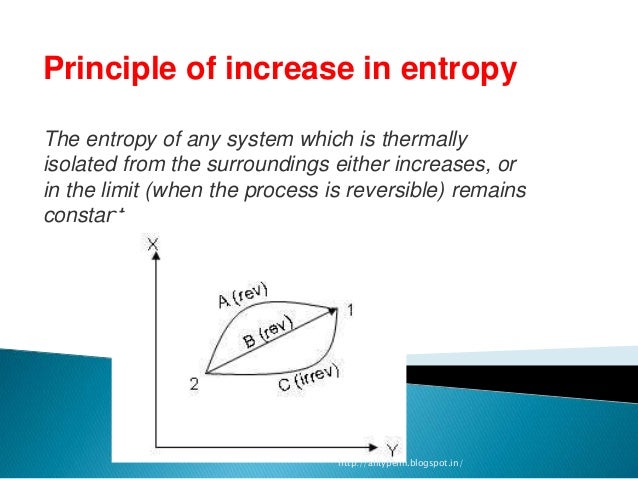
Entropy of a pure perfect crystalline substance is zero at absolute zero. Thermodynamics deals with the relation between that small part of the universe in which we are interested - the system - and the rest of the universe - the surroundings. A perfect crystal at absolute temperature i.e., 0 K, is in state of minimum entropy because each atom is fixed at a particular position in the crystal lattice. Entropy is dynamic - something which static scenes dont reflect. At STP conditions, the state of a substance is the most stable, also called standard state.Įntropy can be defined as a measure of degree of randomness or disorder in a system or it can also be thought as a relative distribution of the energy within the system. Absolute entropy, also known as the entropy at standard conditions, refers to the entropy content of a substance at a standard state. Earhart 2 of 2 All standard state, 25 C and 1 bar (written to 1 decimal place). Where S° is the standard entropy change of the reaction. At any temperature over 0°K, the conventional entropies of all substances, whether elements or compounds, are always positive. For energy dispersal at 0 K: Energy is as concentrated as it can be. The only way is if all species have the same absolute entropy at 0 K. 
This shows that the entropy of the two crystalline forms are the same. Recall the standard temperature and pressure conditions, also called STP conditions. The standard entropy (S°) is the absolute entropy of a pure material at 25☌ (298 K) and 1 atm pressure. Schematic showing the entropy of the rhombic and monoclinic structures of sulfur as a function of temperature. Hint: Here, in the question standard temperature (in K) is asked at which standard absolute entropy exists.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
